Serum Origin Definitions
The ISIA has developed definitions of origin based on international guidelines set by the WTO, OIE, USDA, and the EU — providing precise wording to avoid confusion across the global serum supply chain.
Serum origin definitions
The ISIA has developed definitions of origin based on international guidelines set by the WTO, OIE, USDA, and the EU. Since the word "origin" has several meanings and can cause confusion and controversy, ISIA suggests the use of very precise wording whenever possible.
The three key terms — Country of Collection, Country of Manufacturing, and Country of Further Treatment — each carry a distinct meaning that must be accurately documented on the Certificate of Analysis.
Official ISIA Origin Definitions documentCountries of origin
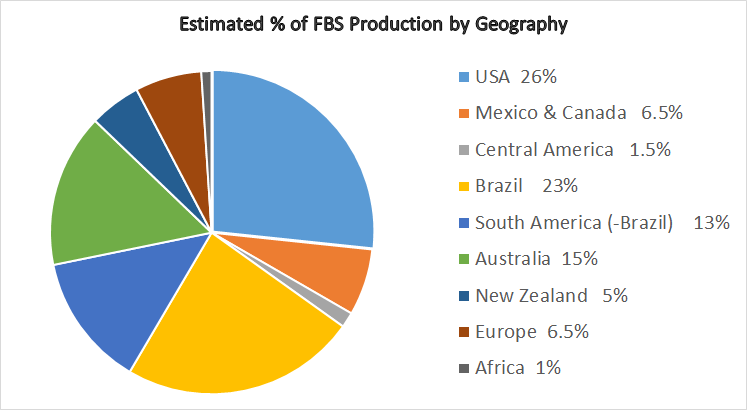
The knowledge of what a material is and where it was sourced is the core of the ISIA Traceability Program, and is critical to the credibility of the industry with customers and regulators. Almost the first action of the Association after it was formed was to agree on the definition of "origin" — currently defined as the location where the animal was killed or, in the case of donor animals, where the blood was collected.
According to the ISIA Traceability Program, Certificates of Origin or Analysis must reflect all origins present, in descending order of prevalence. ISIA strongly discourages the use of mixtures of sources except in specific instances such as Central American serum.
ISIA actively discourages the use of misnomers such as "USDA Approved", "USDA Grade", "EU Approved", or "EU Grade".